One of the first patients in the South West treated with ‘life changing’ Zolgensma on the NHS
Last updated: 06/07/2021
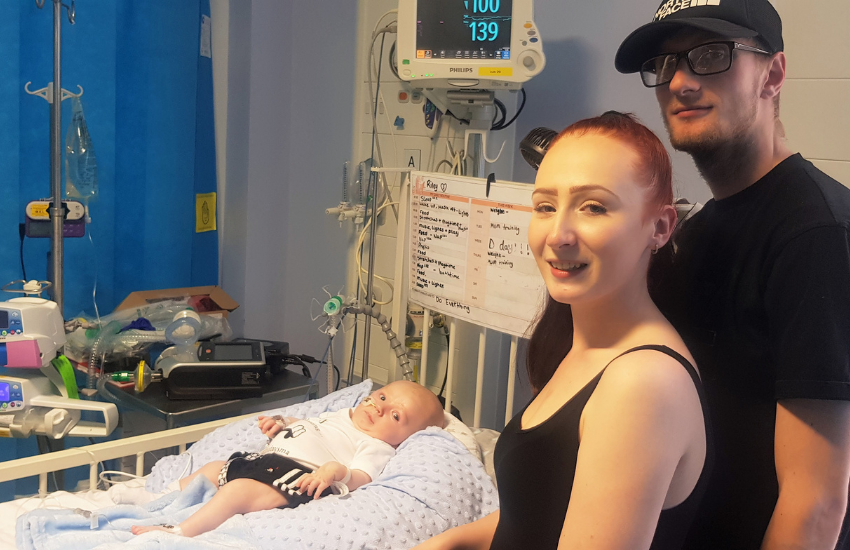
A three month old baby boy has become one of the first patients in the south west of England to receive Zolgensma on the NHS, a life-changing drug that can enable mobility and prolong life in babies and young children with Spinal Muscular Atrophy (SMA) Type 1.
Riley Cadle-Birch, who was diagnosed with SMA Type 1 when he was just four weeks old, became one of the first patients to receive the one-off gene therapy at Bristol Royal Hospital for Children on 30 June.
Zolgensma, dubbed the world’s most expensive drug with an NHS list price of £1.79 million per dose, is now available to patients on the NHS following a landmark deal struck in March.
The one hour long intravenous infusion works by providing a functional copy of the gene known as SMN1, which the body needs to make a protein that is essential for the normal functioning of nerves which control muscle movements. Patients with SMA have a defective copy of this gene, so when Zolgensma is delivered through an intravenous infusion, it passes into the nerve cells and provides instructions to the body to produce the protein and thereby restore nerve function.
Babies born with severe type 1 SMA – the most common form of the condition – experience paralysis, muscle weakness and progressive loss of movement and have a life expectancy of just two years.
In studies, Zolgensma, manufactured by Novartis Gene Therapies, has helped babies to reach milestones such as breathing without a ventilator, sitting up on their own and crawling and walking after the single infusion treatment.
University Hospitals Bristol and Weston NHS Foundation Trust (UHBW), which includes Bristol Royal Hospital for Children, was announced as one of four centres across the country which have been commissioned to administer the gene therapy.
The other three sites which have been confirmed are Manchester University NHS Foundation Trust, Sheffield Children’s NHS Foundation Trust, and Evelina London Children’s Hospital (part of Guy’s and St Thomas’ NHS Foundation Trust) and with agreement to develop a sub hub at Great Ormond Street Hospital for Children NHS Foundation Trust.
Riley underwent the gene therapy infusion last week after responding well to another form of treatment for SMA, nusinersen (also known as Spinraza).
His mother, Jade Cadle-Billingham, first noticed that something was wrong with Riley’s hand. This was also picked up by their midwife during an appointment when Riley was three weeks old, along with some issues with his breathing.
Riley was taken to hospital for a series of tests to check his reflexes, and was later diagnosed with SMA Type 1.
After a difficult start, with a period of time spent on life support, Riley begun receiving Spinraza. This had a positive effect on Riley, so his medical team at Bristol Royal Hospital for Children discussed his case at the national Zolgensma multi-disciplinary meeting with the other three centres, and it was agreed that Riley met the criteria for receiving this life-changing gene therapy treatment.
Jade, from Gloucester, said: “We’ve been on a really rocky road with Riley, with him being so ill at times we weren’t sure if he would make it.
“When we started to suspect he may have SMA, I began researching the condition and came across Zolgensma, which at the time had only been announced as approved by the NHS, but we didn’t yet know where it would be available or if Riley would be able to have it.
“We want to do anything we can to raise awareness of this life-changing treatment, and to give hope to other SMA families going through the same thing.
“Having this treatment is like giving him his future.”
Dr Kayal Vijayakumar, consultant paediatric neurologist at Bristol Royal Hospital for Children and clinical lead for the hospital’s neuromuscular service, said:
“I feel very privileged as part of this team to be in a position where we can now offer families this ground breaking treatment. The data from scientific studies illustrates that this treatment can significantly change the lives of those children affected by Spinal Muscular Atrophy.
“We’re incredibly excited to have been chosen as one of the four national centres in England, and look forward to working closely with the other centres to ensure eligible patients are able to receive treatment regardless of where they live.
“I hope the success of this treatment will act as a springboard for more gene therapies to become available in the future.”
Jon Standing, Director of Pharmacy at University Hospitals Bristol and Weston NHS Foundation Trust said
“It’s a privilege for us as a pharmacy department to be involved in the preparation and provision of this ground breaking treatment. It will be a true team approach with many services in the Trust pulling together to deliver the service.
“This treatment offers real hope of a brighter future for SMA Type 1 patients and their families.”